Bacteria in a new hospital’s drinking water system: reasons and recommended renovations
7-minute read
Sanitary fittings can be endangered by upstream sources of contamination. Finding and fixing the cause of this contamination is necessary for the success of the renovation project. One particular hazard arises in cases where the bacterium Pseudomonas aeruginosa is discovered shortly before system commissioning, since this can be a health hazard in certain circumstances.
During initial inspection of a new hospital development, elevated ‘general colony counts’ were detected at some tapping points and Pseudomonas aeruginosa found on analysis. Since routine sampling according to DIN ISO 19458 required only 100 ml to be sampled from the fittings, no conclusions could be drawn about potential contamination of the upstream installation. Suitable sampling points and methods now needed to be selected.
Pseudomonas aeruginosa
Key goals of the investigation
The most important standard governing the microbiological sampling of drinking water installations is DIN EN ISO 19458 (‘Water quality – Sampling for microbiological analysis’). The standard sets out three purposes here.
- The first of these, a), is to evaluate the water from the utility to the water meter. Accordingly, the supply line must always be thoroughly flushed before sampling.
- The second, b), conducts tests to verify that high-quality drinking water from the utility is present at the tapping fittings. Accordingly, sampling is conducted without flow regulators or a shower hose: the outlet area is disinfected and a small volume is discarded. This is necessary because these components are often a place where bacteria congregate – these may have originated in air or on utensils such as washing up or cleaning cloths and can falsify results.
- The final purpose, c), involves sampling the water as it leaves the fitting – namely via the flow regulator or shower hose – with no prior disinfection and without letting water run first. Tests conducted for purpose c) can clarify whether users could have acquired an infection when using water taken from this fitting, for example.

Is contamination local or systemic?
Preserving water quality in buildings is based on the regular exchange of water across all tapping points. Temperature control is similarly performed in the installation, with cold water and hot water both being regularly piped up to the tapping points. The water that then flows from the installation must be microbiologically safe. For this reason, it is important to test it using a staged approach to sampling.
If the drinking water from a utility line is sound, but the sample taken according to ISO 19458 purpose b) is not, then one may assume that the fitting itself is contaminated. In this case, three basic causes could be relevant, which need to be clarified using sampling:
Cause 1 – were the fittings and other components already contaminated when installed?
Cause 2 – have the fittings and components been contaminated by the upstream installation?
Cause 3 – have the tapping points been contaminated by the flow regulator, because no adequate exchange of water took place here?
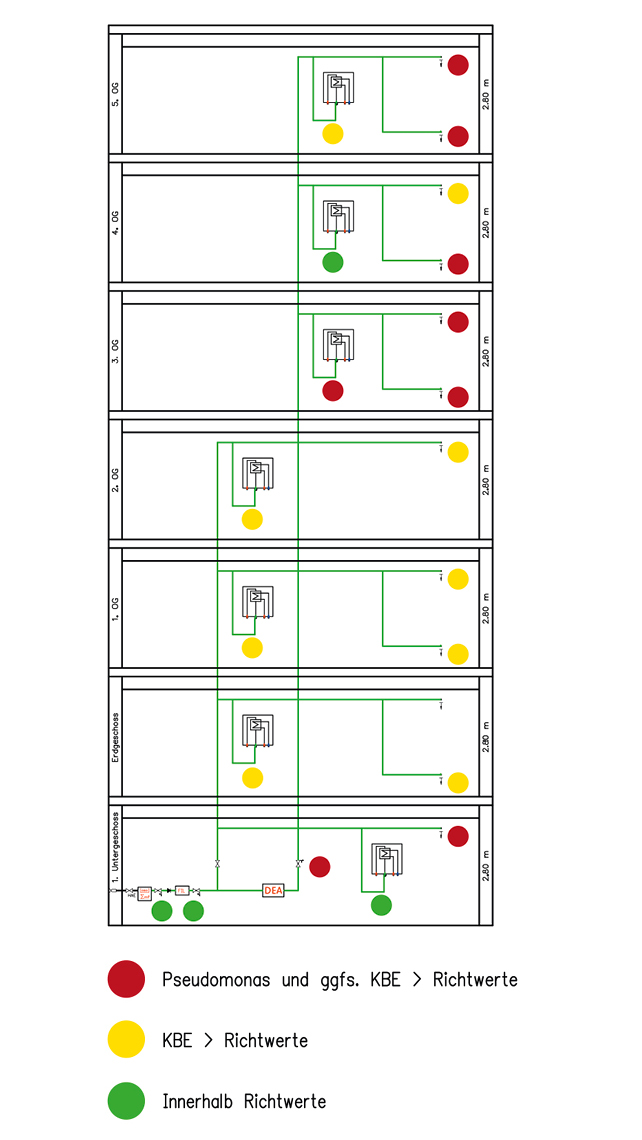
Upstream contamination as a cause
In the hospital concerned, the cause of contamination was determined using a systematic analysis of findings and their assignment to the various sections of the installation. When comparing data from floors with a pressure boosting station (floors 3, 4 and 5) and without a pressure boosting station (basement, ground floor, floors 1 and 2), it was found that Pseudomonas aeruginosa had been detected only on the floors supplied with a pressure boosting station. While colony counts were indeed elevated on the floors below, no Pseudomonas bacteria were present. Since the same fittings had been installed there as on the contaminated floors, one could therefore conclude that the fittings on floors 3 to 5 had ‘fallen victim’ to an upstream contamination. This therefore needed to be found in the area from the upstream installation to the pressure boosting station. The pressure boosting station (PBS) installed consisted of two pumps, operated alternately. These were sampled, resulting in the sporadic detection of Pseudomonas aeruginosa. With the contaminated PBS confirmed, the cause had been found, along with the most important starting-point for a rapid and effective clean-up. The PBS and the entire installation were subjected to intensive flushing over two months and thermally disinfected section by section across all tapping points. Hygienic optimisations were also made to the operation of the PBS by reducing the switchover intervals of the two pumps to 60 minutes.

Electronic fittings – here the XERIS E-T with thermostat – can automatically trigger a water exchange during a usage interruption.
Recommendations for practice
Components tested with water at the factory cannot always be avoided during procurement. However, design planners should ensure that the tender documents specify dry testing on the part of the manufacturer – as is done at SCHELL – for as many parts as possible. If such parts are not available, then manufacturers should take appropriate steps and offer instructions on hygiene-compliant handling. For buildings with more stringent hygienic requirements, it is also recommended to complete installation filling section by section, with each of these steps accompanied by microbiological sampling and approval.
The modern trend is towards an increased deployment of electronic fittings in buildings with more stringent hygienic requirements, so as to ensure an automated exchange of water even during interruptions in use. In this way, the fittings help to ensure optimum water quality, cost-effective operations and legal compliance for operators.

























![[Translate to English:] [Translate to English:]](/fileadmin/user_upload/images/menu/menu_service_downloads_broschueren.jpg)






![[Translate to English:] [Translate to English:]](/fileadmin/_processed_/7/7/csm_menu_unternehmen_ueber-schell_awards_f6cec25b1d.jpg)
![[Translate to English:] [Translate to English:]](/fileadmin/_processed_/a/0/csm_menu_unternehmen_ueber-schell_wasser-sparen_41036d2dd9.jpg)